Biological Engineering & Molecular Innovation
Strain Development
Industrial-ready chassis engineered for scalable biosynthesis
We integrate multiple biosynthetic systems—including microbial cell factories, enzymatic catalysis, and cell-free systems—to identify the optimal biomanufacturing route for each target molecule.
Supported by AI-driven computational design and automated microfluidic high-throughput screening, our platform enables rapid iteration, performance optimization, and early-stage industrial validation—significantly accelerating the path from laboratory development to large-scale manufacturing.

Enzyme Design
Precision-engineered enzymes for efficient and robust catalysis
Our team leverages decades of expertise in enzyme catalysis, focusing on novel enzyme discovery, substrate expansion, and activity enhancement.
By integrating enzyme kinetics, structural biology, and protein engineering, we systematically design and optimize key catalytic enzymes. Powered by AI-driven computational platforms and automated high-throughput screening, we develop highly stereoselective enzymes with exceptional efficiency and extended operational stability—providing a robust foundation for high-performance biosynthetic pathways.
Metabolic Pathway Optimization
System-level control for cost-defining biosynthesis
We apply advanced genome-editing toolkits across diverse chassis systems to rapidly convert wild-type strains into high-performance production hosts. Modular genetic design enables efficient pathway transfer and reproducible expression across multiple hosts, supporting flexible multi-molecule development.
Guided by an omics-driven diagnostic framework, we systematically analyze changes in intracellular material and energy fluxes, enabling the rational design of sophisticated metabolic control circuits. Through the modular orchestration of multiple metabolic pathways and essential cofactors, we establish flexible optimization packages that can be rapidly reconfigured to accelerate strain development. This allows precise balancing between growth and production, maximizing pathway efficiency while maintaining robust cellular performance. Beyond short-term titer improvement, our optimization strategy prioritizes long-term strain stability, scalability, and process consistency under industrial fermentation conditions—ultimately driving significant unit cost reduction and reliable commercial-scale manufacturing.

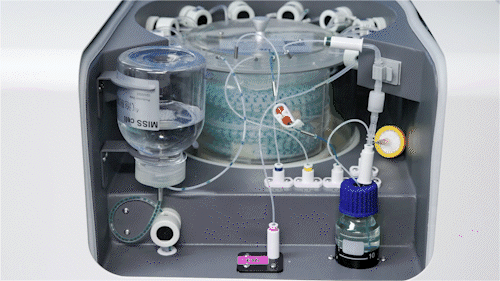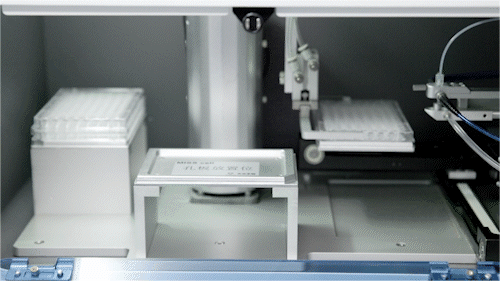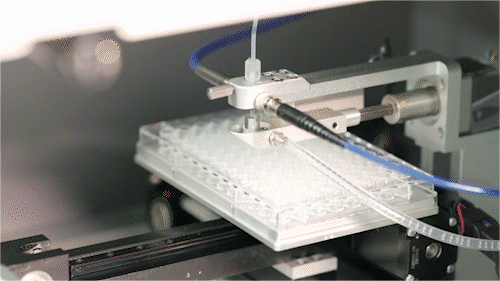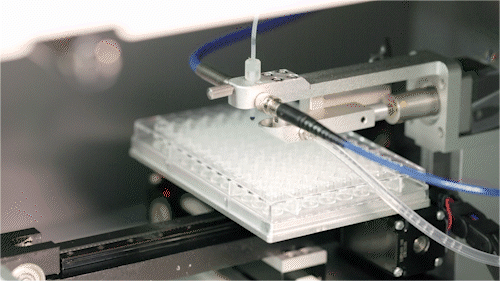
Automated Microfluidic Screening
We have established a fully integrated microfluidic high-throughput screening platform spanning the DNA/RNA–protein–cell continuum, seamlessly coupled with automated robotics to enable a closed-loop workflow from construct generation and screening to data acquisition.
Operating across multiple screening dimensions, the platform achieves throughput of up to millions of cells per hour, enabling rapid identification of high-performing enzymes and engineered strains with optimal catalytic efficiency, metabolic performance, and operational stability. Continuous accumulation of high-quality, multi-dimensional datasets further powers AI model training and predictive design, accelerating the Design–Build–Test–Learn (DBTL) cycle and overall R&D efficiency.